 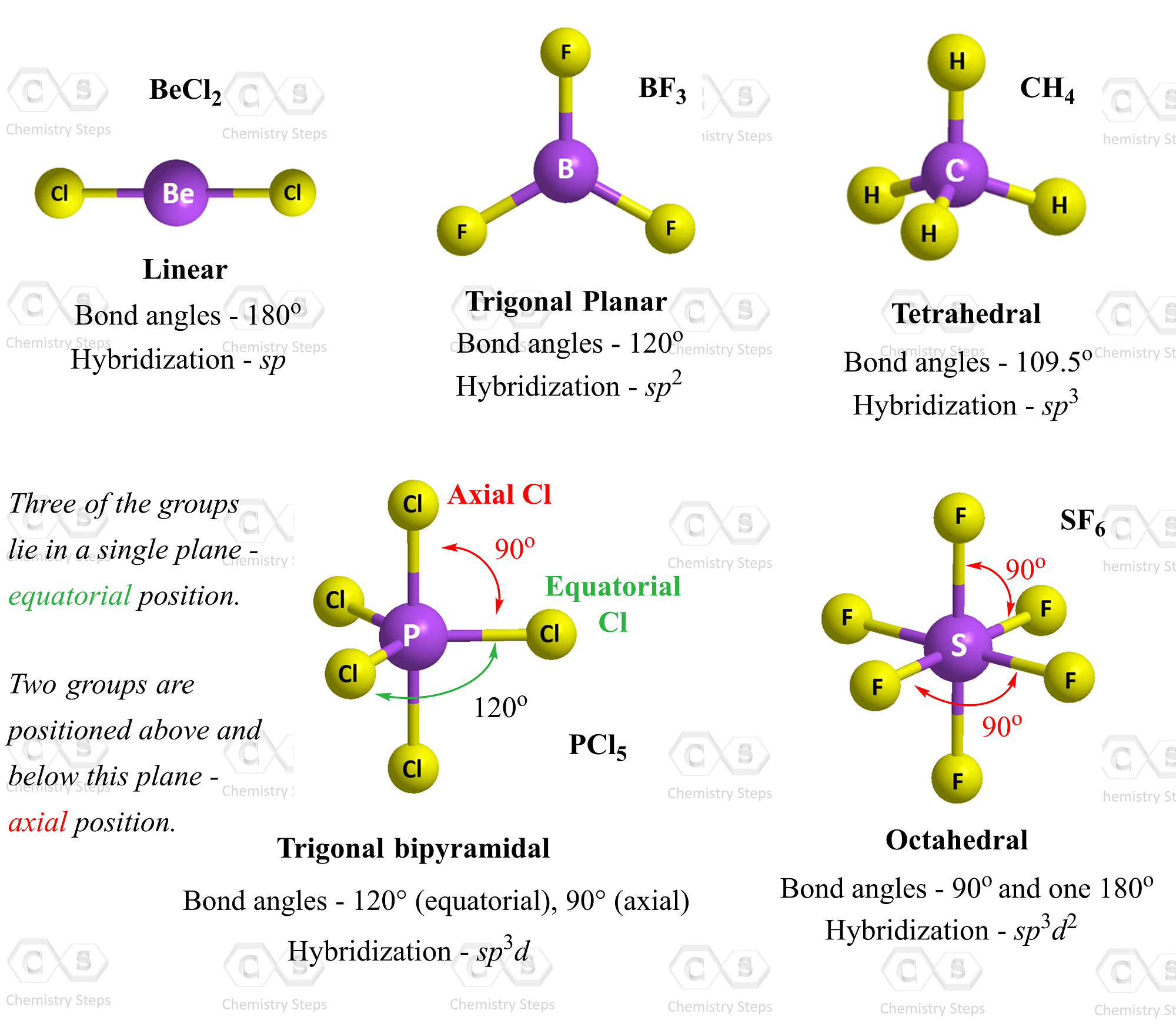
X as the atoms or ligands attached to the central atom Į as the non-bonding electron pairs/lone pairs Here are all the different combinations of molecular geometries with lone pairs: One easy tip in determining your final structure is to set up your electron clouds in your default geometry (including your lone pairs), and then ignore the lone pairs to determine the final shape. The reason why lone pairs make such a difference in your geometry is because the lone pairs take up more space, and thus compress the bond angles of the terminal atoms. To determine your final molecular geometry, you must count the number of lone pairs in your molecule, and consider what the default geometry of your molecule is. Molecular Geometry (inlcuding the Lone pairs) Now, as I have stated before, this just gives you your default geometry you must further assess the lone pairs in your molecule to get the final geometry that you will state as your answer. ** Note – this is less commonly discussed in secondary/first year studies of molecular geometry Seven electron clouds = pentagonal bipyramidal (bond angle: 72º and 90º) **.Six electron clouds = octahedral (bond angle: 90º and 180º). 
Five electron clouds = trigonal bipyramidal (bond angle: 90º and 180º).Four electron clouds = tetrahedral (bond angle: 109.5º).Three electron clouds = trigonal planar (bond angle: 120º). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
